
Clinical Trials for Pain Management sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality. From defining clinical trials to exploring ethical considerations, this overview covers the key aspects of this crucial medical research field.
Introduction to Clinical Trials
Clinical trials are research studies conducted on human volunteers to evaluate the safety and effectiveness of new medical treatments, interventions, or drugs. These trials play a crucial role in advancing medical knowledge and improving patient care.
Importance of Clinical Trials in Advancing Pain Management Treatments
Clinical trials are essential in the field of pain management as they help researchers determine the efficacy of new treatments in relieving pain and improving the quality of life for patients. By testing new drugs or interventions in a controlled setting, clinical trials provide valuable data that can lead to the development of more effective pain management strategies.
- Clinical trials help identify the most promising treatments for pain relief by comparing them to standard care or a placebo.
- These trials also help researchers understand the potential side effects and risks associated with new treatments, ensuring patient safety.
- Through clinical trials, researchers can gather evidence to support the approval of new pain medications by regulatory authorities.
Phases of Clinical Trials and their Significance in Drug Development
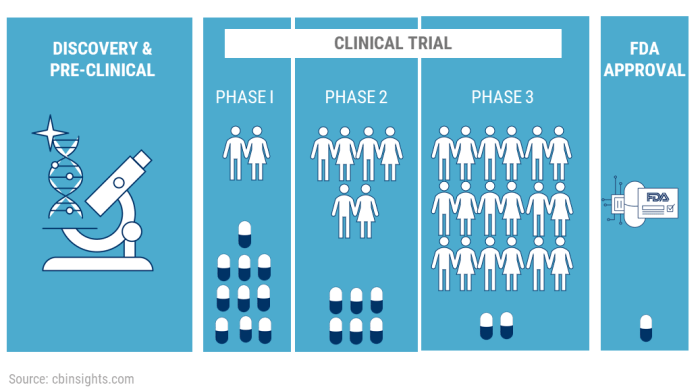
Clinical trials are typically divided into four phases, each serving a specific purpose in the drug development process.
- Phase 1: In this phase, the focus is on evaluating the safety and dosage of the new treatment in a small group of healthy volunteers. This phase helps researchers determine how the drug is metabolized and excreted in the body.
- Phase 2: During this phase, the treatment is tested on a larger group of patients to assess its effectiveness and side effects. Researchers aim to gather preliminary data on the drug’s efficacy in treating pain.
- Phase 3: In this phase, the treatment is compared to existing standard treatments or a placebo in a larger population to further evaluate its effectiveness, safety, and optimal dosage. This phase provides crucial evidence for regulatory approval.
- Phase 4: This phase occurs after the treatment has been approved and is on the market. Researchers continue to monitor its safety and effectiveness in real-world settings, gathering long-term data on its benefits and risks.
Types of Clinical Trials for Pain Management
Clinical trials for pain management can vary in their design and purpose. Understanding the different types of clinical trials is essential for evaluating the effectiveness of treatments and interventions.
Randomized Controlled Trials (RCTs)
Randomized controlled trials are considered the gold standard in clinical research. In RCTs, participants are randomly assigned to different groups: one receiving the treatment being tested and the other receiving a control treatment or a placebo. This design helps to minimize bias and determine the true effects of the treatment.
Observational Studies
Observational studies, on the other hand, do not involve any intervention by the researchers. Instead, researchers observe participants over time to gather data on the natural progression of the disease or condition. These studies are useful for identifying associations between risk factors and outcomes.
Interventional Clinical Trials
Interventional clinical trials involve actively administering a treatment or intervention to participants to assess its effectiveness. These trials often include randomized controlled trials but can also involve other study designs, such as crossover trials or adaptive trials.
Observational Clinical Trials
Observational clinical trials, as mentioned earlier, do not involve interventions by the researchers. Instead, they focus on observing participants in real-world settings to gather data on the safety and effectiveness of treatments. These trials are valuable for assessing the long-term outcomes of treatments.
Differences between Interventional and Observational Trials
The main difference between interventional and observational trials lies in the level of control researchers have over the treatment. In interventional trials, researchers actively administer treatments, while in observational trials, researchers observe participants without intervening. Both types of trials play a crucial role in advancing our understanding of pain management and improving patient outcomes.
Participant Recruitment and Selection

Participant recruitment and selection are crucial aspects of pain management clinical trials to ensure the efficacy and safety of the treatments being studied.
Recruitment Process
Participants for pain management clinical trials are typically recruited through various channels, including:
- Medical centers and hospitals
- Community outreach programs
- Online platforms and social media
Selection Criteria
When selecting participants for clinical trials on pain management, researchers consider several criteria to ensure the study’s validity and safety:
- Age: Participants must fall within a specific age range to ensure the treatment’s effectiveness across different age groups.
- Gender: Some trials may require a specific gender distribution to study potential gender-specific effects of the treatment.
- Medical History: Participants with a history of certain conditions or treatments may be excluded to avoid confounding variables.
- Pain Severity: Participants are usually selected based on the severity of their pain to assess the treatment’s impact on different pain levels.
Importance of Diversity
Diversity in participant selection is crucial for pain management clinical trials for several reasons:
- Effectiveness Assessment: A diverse participant pool helps researchers understand how treatments work across different demographics, ensuring broader applicability.
- Safety and Side Effects: Diverse groups can help identify potential side effects or safety concerns that may vary among different populations.
- Ethical Considerations: Ensuring diversity in participant selection promotes fairness and equity in research practices, avoiding biases or exclusions.
Ethical Considerations in Clinical Trials
Clinical trials for pain management are governed by strict ethical guidelines to ensure the safety and well-being of participants. These guidelines are put in place to protect the rights of individuals involved in research studies and to uphold the principles of beneficence, non-maleficence, autonomy, and justice.
Informed Consent Procedures
Informed consent is a crucial aspect of ethical clinical trials. Before participating in a study, participants must be fully informed about the purpose of the research, potential risks and benefits, procedures involved, and their rights as a participant. This ensures that individuals can make an informed decision about whether or not to take part in the trial.
Protection of Participants’ Rights
Participants’ rights are of utmost importance in clinical trials. These rights include the right to privacy, confidentiality, voluntary participation, and the right to withdraw from the study at any time without facing any consequences. Researchers must ensure that these rights are respected throughout the course of the trial.
Role of Institutional Review Boards (IRBs)
Institutional Review Boards play a vital role in ensuring ethical conduct during clinical trials. IRBs are independent committees comprised of medical professionals, researchers, ethicists, and community members who review and approve research studies to ensure that they meet ethical standards. IRBs carefully assess the risks and benefits of the study, the informed consent process, and the protection of participants’ rights before granting approval for the trial to proceed.
Data Collection and Analysis
Data collection and analysis are crucial components of pain management clinical trials. Researchers employ specific methods to collect data and analyze it to draw conclusions about the effectiveness of treatments.
Data Collection Methods
In pain management clinical trials, data is typically collected through various means such as patient-reported outcomes, physical examinations, laboratory tests, imaging studies, and electronic health records. These methods help researchers gather comprehensive information about the participants’ pain levels, treatment responses, and overall health status.
Data Analysis Process
Once the data is collected, researchers analyze it using statistical methods to identify patterns, trends, and correlations. They compare the outcomes of different treatment groups to determine the effectiveness of the interventions. By analyzing the data, researchers can draw conclusions about the impact of the treatments on pain relief and overall patient well-being.
Importance of Data Integrity
Data integrity is essential in clinical trials to ensure the accuracy and reliability of the results. Researchers must maintain strict protocols to prevent data manipulation or bias, which could compromise the validity of the findings. By upholding data integrity, researchers can uphold the credibility of the study outcomes and provide reliable evidence to support clinical decision-making in pain management.
Challenges and Limitations in Pain Management Clinical Trials
Clinical trials for pain management face several challenges and limitations that can impact the validity of the study outcomes. It is essential to address these issues to ensure the reliability and effectiveness of pain management strategies.
Common Challenges Faced in Conducting Clinical Trials for Pain Management
- Inadequate participant recruitment: Difficulty in finding suitable participants can lead to delays in the trial process.
- High dropout rates: Participants may drop out of the study due to adverse effects or lack of efficacy, affecting the data analysis.
- Placebo effect: The placebo effect can influence the perceived effectiveness of the treatment being tested.
- Heterogeneity of pain conditions: Pain is a complex and subjective experience, making it challenging to standardize treatments across different types of pain.
Limitations of Clinical Trials in Pain Management
- Small sample sizes: Limited sample sizes can reduce the statistical power of the study and affect the generalizability of the results.
- Bias: Bias in participant selection, data collection, or analysis can impact the accuracy and reliability of the findings.
- External factors: External variables, such as environmental influences or individual differences, can confound the results of the clinical trial.
Ways to Overcome These Challenges and Improve Validity
- Implement rigorous participant screening criteria to ensure the selection of suitable candidates for the trial.
- Utilize randomized controlled trials with double-blind designs to minimize bias and placebo effects.
- Collaborate with multidisciplinary teams to address the heterogeneity of pain conditions and tailor treatments accordingly.
- Conduct multi-center trials to increase sample sizes and enhance the generalizability of the results.
In conclusion, Clinical Trials for Pain Management play a vital role in shaping the future of pain treatment. By understanding the phases, challenges, and ethical guidelines, researchers can continue to innovate and improve patient care. Stay informed and engaged in this dynamic field to contribute to advancements in pain management.
Questions and Answers
How are participants recruited for clinical trials?
Participants are recruited through various methods, such as advertising, medical referrals, or patient databases.
What are the different types of clinical trials for pain management?
Types include randomized controlled trials, observational studies, and more, each serving different research purposes.
Why is diversity important in participant selection for clinical trials?
Diversity ensures that results are applicable to a broader population, improving the effectiveness of treatments.




