
Clinical Trials for New Medications sets the stage for this enthralling narrative, offering readers a glimpse into a story that is rich in detail and brimming with originality. From explaining the purpose of clinical trials to exploring the significance of data collection and analysis, this topic delves into the fascinating world of medical research and innovation.
CLINICAL TRIALS
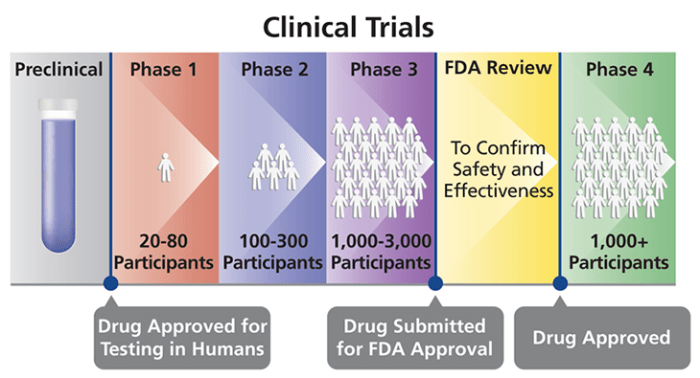
Clinical trials play a crucial role in the development and testing of new medications. These trials are essential to ensure the safety and efficacy of the drugs before they are made available to the general public.
Different Phases of Clinical Trials
- Phase 1: This phase involves a small group of healthy volunteers to test the safety and dosage of the medication. The focus is on understanding how the drug is metabolized and its potential side effects.
- Phase 2: In this phase, a larger group of patients who have the condition the medication is designed to treat is involved. The goal is to further evaluate the drug’s safety and effectiveness.
- Phase 3: This phase includes a much larger group of patients to confirm the drug’s effectiveness, monitor side effects, and compare it to existing treatments. This phase provides crucial data for regulatory approval.
Each phase of a clinical trial serves a specific purpose in assessing the safety and efficacy of a new medication.
Importance of Participants’ Informed Consent
Clinical trial participants must provide informed consent before joining a study. This means they have been fully informed about the risks, benefits, and procedures involved in the trial. Informed consent ensures that participants understand what they are agreeing to and have the autonomy to make decisions about their participation.
Participants’ informed consent is crucial in upholding ethical standards and respecting the rights and well-being of individuals involved in clinical trials.
NEW MEDICATIONS

Developing new medications involves a rigorous process that typically starts with extensive research and testing in laboratories. Scientists work to identify potential compounds that could have therapeutic effects on specific diseases or conditions.
Selection for Clinical Trials
Once a promising compound is identified, it goes through preclinical testing to evaluate its safety and efficacy. If the results are favorable, the medication may be selected for clinical trials.
- Potential new medications are carefully evaluated based on their mechanism of action, safety profile, and potential benefits for patients.
- Researchers consider factors such as the severity of the disease being targeted, the availability of existing treatment options, and the feasibility of conducting clinical trials.
- Selection criteria for clinical trials are designed to ensure that the medication is tested in populations that are most likely to benefit from it.
Role of Regulatory Bodies
Regulatory bodies play a crucial role in overseeing the testing of new medications to ensure patient safety and the reliability of the data generated during clinical trials.
- Regulatory agencies, such as the Food and Drug Administration (FDA) in the United States, review the data from preclinical studies and clinical trials to determine whether a new medication is safe and effective for use.
- These agencies set standards for the design and conduct of clinical trials, monitor the progress of studies, and ultimately decide whether a new medication can be approved for market authorization.
- Regulatory bodies work to balance the need for timely access to new treatments with the importance of ensuring that medications meet high standards of quality, safety, and efficacy.
PARTICIPANTS IN CLINICAL TRIALS
Clinical trials are essential for testing the safety and efficacy of new medications before they are available to the general public. The selection of participants in these trials is a crucial process that involves certain criteria to ensure the validity of the results.Potential participants in clinical trials are usually selected based on specific criteria such as age, gender, medical history, and the condition being studied.
These criteria help researchers to identify individuals who are most likely to benefit from the medication being tested and also ensure the safety of the participants.
Criteria for Selecting Participants
- Participants must meet certain age and health requirements to ensure they are suitable for the study.
- Individuals with specific medical conditions or characteristics related to the study are often sought after.
- Participants must be willing to follow the study protocol and attend scheduled appointments.
Risks and Benefits of Participation
- Participants in clinical trials may experience side effects or adverse reactions to the medication being tested.
- On the other hand, participants have the opportunity to access potentially life-saving treatments before they are widely available.
- Being part of a clinical trial can also provide valuable medical monitoring and support from healthcare professionals.
Importance of Diversity in Participants
- Diversity in clinical trial participants is crucial to ensure that the results are representative of the general population.
- Including individuals from different backgrounds, ethnicities, and geographical locations helps to understand how the medication may affect various groups.
- It is important to have diverse participants to avoid biases and ensure that the medication is safe and effective for everyone.
DATA COLLECTION AND ANALYSIS
Data collection during clinical trials is a crucial process that involves gathering information on the participants and the effects of the new medication being tested. Researchers use various methods to collect data, such as patient interviews, medical examinations, laboratory tests, and patient diaries. This data helps in evaluating the safety and efficacy of the medication.
Methods for Data Collection
- Patient interviews: Researchers conduct interviews to gather information about the participants’ medical history, symptoms, and overall health.
- Medical examinations: Physical examinations are performed to monitor any changes in the participants’ health status during the trial.
- Laboratory tests: Blood tests, urine tests, and other laboratory procedures are conducted to assess the impact of the medication on the participants’ body.
- Patient diaries: Participants are often asked to keep diaries to record their symptoms, medication usage, and any side effects experienced.
Data Analysis in Clinical Trials
Data collected during clinical trials are carefully analyzed and interpreted by researchers to determine the effectiveness and safety of the new medication. Statistical methods are commonly used to analyze the data, comparing outcomes between the treatment group and the control group. The results are then used to draw conclusions about the medication’s benefits and risks.
Significance of Data Integrity and Accuracy
- Data integrity is crucial in ensuring the reliability of the clinical trial results. Any inaccuracies or errors in data collection can lead to incorrect conclusions about the medication’s efficacy and safety.
- Accurate data collection is essential for maintaining the credibility of the trial. Researchers must follow strict protocols and guidelines to ensure that the data collected is valid and reliable.
- The outcomes of clinical trials heavily depend on the accuracy of the data collected. It is essential to maintain data integrity throughout the trial to produce meaningful and trustworthy results.
In conclusion, Clinical Trials for New Medications sheds light on the crucial process of testing and developing new medications. By understanding the phases of clinical trials, the selection of participants, and the importance of data integrity, we gain insight into the intricate journey of bringing innovative treatments to the forefront of healthcare.
Clarifying Questions
What is the purpose of clinical trials?
Clinical trials aim to test the safety and effectiveness of new medications.
How are potential new medications selected for clinical trials?
Potential new medications are selected based on preclinical research and their potential benefits.
Why is diversity important in clinical trial participants?
Diversity ensures that results are representative across different demographics, improving the generalizability of the findings.
How do researchers analyze data from clinical trials?
Researchers use statistical methods to analyze and interpret the data collected during clinical trials.
What role do regulatory bodies play in overseeing new medication testing?
Regulatory bodies ensure that clinical trials are conducted ethically and that new medications meet safety standards before approval.





