
Embark on a journey through the realm of Clinical Trial Results and Findings, where the discoveries and implications of medical studies come to light in a captivating narrative.
Delve into the core aspects of clinical trials, their significance, and the crucial role they play in shaping future healthcare practices.
CLINICAL TRIALS
Clinical trials are research studies conducted to evaluate the safety and effectiveness of new treatments, interventions, or drugs before they are approved for widespread use. These trials play a crucial role in advancing medical knowledge and improving patient care by providing evidence-based data on the benefits and risks of various medical interventions.
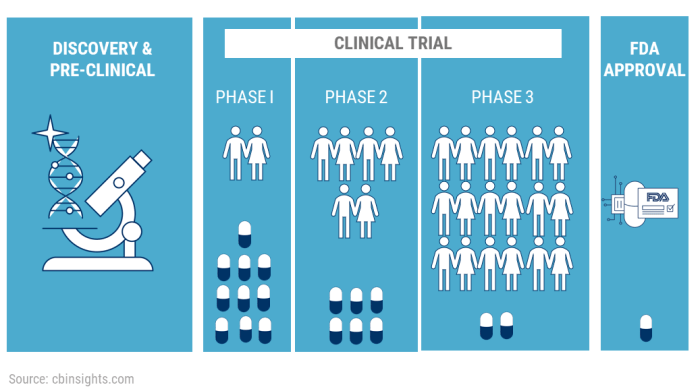
Phases of Clinical Trials
Clinical trials are typically conducted in four phases, each with specific objectives and procedures:
- Phase 1: Involves a small group of healthy volunteers to assess the safety, dosage, and side effects of the treatment.
- Phase 2: Expands to a larger group of patients to determine the treatment’s effectiveness and further evaluate its safety.
- Phase 3: Involves a larger population to confirm the treatment’s effectiveness, monitor side effects, and compare it to existing treatments.
- Phase 4: Occurs after the treatment is approved and involves continued monitoring in real-world settings to gather additional information on its safety and effectiveness.
Informed Consent in Clinical Trials
Informed consent is a critical component of ethical clinical research, ensuring that participants fully understand the study’s purpose, risks, benefits, and procedures before deciding to enroll. It involves providing detailed information in a language that participants can comprehend, allowing them to make an informed decision about their participation. Without informed consent, clinical trials would lack ethical integrity and pose risks to participants’ autonomy and well-being.
CLINICAL TRIAL RESULTS AND FINDINGS
When it comes to clinical trial results and findings, publishing them in scientific journals is of utmost importance. This allows for transparency, peer review, and dissemination of crucial information to the medical community and the public.
Researchers analyze and interpret clinical trial findings through rigorous statistical methods and data analysis techniques. They look for patterns, correlations, and statistical significance to draw meaningful conclusions from the data collected during the trial.
Impact of Clinical Trial Results
The impact of clinical trial results on future medical treatments and practices cannot be overstated. Positive results can lead to the approval of new drugs or treatments, while negative results can prevent ineffective or harmful treatments from being used in the future. These findings shape the direction of medical research and influence clinical guidelines and best practices.
REPORTING RESULTS
Clinical trial results are typically reported following standard practices to ensure transparency and accuracy in the findings. This involves detailing the outcomes of the study, including both positive and negative results, to provide a comprehensive view of the trial’s findings.
Importance of Transparency and Accuracy
Transparency and accuracy in reporting clinical trial findings are crucial to ensure the credibility and reliability of the results. By providing a clear and honest account of the study outcomes, researchers can uphold ethical standards and contribute to the advancement of medical knowledge.
Reporting Adverse Events
Adverse events observed during clinical trials must be reported accurately to assess the safety and efficacy of the intervention being studied. These events can range from mild side effects to severe complications, and each must be documented and analyzed to determine their impact on the participants’ health.
STATISTICAL ANALYSIS

Statistical analysis is a crucial component of interpreting clinical trial data. It involves using various methods to analyze the results obtained during the trial, helping researchers draw meaningful conclusions from the data.
Methods Used for Statistical Analysis
- Descriptive statistics: This method involves summarizing and presenting the data in a meaningful way, such as mean, median, and standard deviation.
- Inferential statistics: This method helps researchers make inferences or predictions about the population based on the sample data collected during the trial.
- Hypothesis testing: Researchers use hypothesis testing to determine if there is a significant difference between groups in the trial, helping assess the effectiveness of the treatment.
Significance of Statistical Significance
Statistical significance is crucial in interpreting trial results as it indicates whether the observed differences are likely due to the treatment being studied or simply due to chance. A p-value less than 0.05 is commonly used to determine statistical significance, suggesting that the results are unlikely to have occurred by random chance.
Role of Sample Size and Randomization
- Sample size: A larger sample size increases the power of the study, making it more likely to detect a true effect of the treatment. It also helps reduce the margin of error in the results obtained.
- Randomization: Random assignment of participants into treatment groups helps minimize bias and ensures that the groups are comparable at the start of the trial. This helps in drawing accurate conclusions about the effectiveness of the treatment being studied.
In conclusion, the revelations brought forth by Clinical Trial Results and Findings underscore the pivotal role of research in advancing medical knowledge and enhancing patient care.
FAQ Explained
How are clinical trial results published?
Clinical trial results are typically published in scientific journals after undergoing rigorous review processes to ensure accuracy and validity.
Why is statistical analysis important in interpreting trial results?
Statistical analysis helps researchers draw meaningful conclusions from the data collected during clinical trials, providing insights into the effectiveness of treatments.




