
Embark on a journey through Clinical Trials Phases Explained, delving into the intricate phases that shape medical research and drug development.
Explore the nuances of each phase, from Phase 0 to Phase III, and unravel the essential elements that drive clinical trials forward.
CLINICAL TRIALS
Clinical trials are research studies conducted to evaluate the effectiveness and safety of new medical treatments, interventions, or procedures on human volunteers. These trials are crucial in advancing medical knowledge and improving patient care.
Volunteers play a key role in clinical trials by helping researchers gather data to determine the benefits and risks of new treatments. Without the participation of volunteers, it would be impossible to bring new drugs and therapies to the market.
Informed consent is a critical aspect of clinical trials, ensuring that volunteers are fully aware of the potential risks and benefits of participating in the study. This process involves providing detailed information about the trial, including its purpose, procedures, risks, and the participant’s rights, allowing individuals to make an informed decision about their involvement.
Clinical Trials Phases Explained
Clinical trials are conducted in different phases to ensure the safety and efficacy of new drugs or treatments before they are approved for public use. Each phase serves a specific purpose and has unique objectives that contribute to the overall understanding of the treatment being studied.
Phase I Clinical Trials
Phase I clinical trials are the first step in testing a new treatment in humans. The primary objective of Phase I trials is to determine the safety and dosage of the treatment. These trials typically involve a small group of healthy volunteers who are closely monitored for any adverse effects. Researchers use the data gathered from Phase I trials to establish the initial safety profile of the treatment and identify the maximum tolerated dose.
Criteria for Advancing to the Next Phase
To move from one phase to the next in clinical trials, specific criteria must be met. These criteria often include the safety and tolerability of the treatment in the previous phase, as well as any preliminary evidence of efficacy. Regulatory authorities closely review the data from each phase before granting permission to proceed to the next stage. It is essential to demonstrate that the benefits of advancing to the next phase outweigh the potential risks for participants.
Phase 0 Clinical Trials

Phase 0 clinical trials, also known as exploratory investigational new drug (eIND) studies, are the initial step in the drug development process. These trials involve administering a very small dose of the investigational drug to a small number of subjects to gather preliminary data on how the drug is metabolized and how it behaves in the human body.
Goals and Objectives of Phase 0 Trials
- Assessing how the investigational drug is metabolized and distributed in the body.
- Evaluating the drug’s pharmacokinetics and pharmacodynamics.
- Determining if the drug reaches the intended target tissue.
- Establishing a safe starting dose for subsequent clinical trials.
Examples of When Phase 0 Trials Are Utilized
- Investigating drug-drug interactions.
- Exploring the feasibility of a new drug delivery method.
- Testing the drug’s mechanism of action in humans.
- Screening multiple drug candidates to prioritize for further development.
Key Differences Between Phase 0 and Phase I Trials
- In Phase 0, a sub-therapeutic dose is given to a limited number of subjects, while Phase I involves escalating doses in a larger group.
- Phase 0 trials focus on early pharmacokinetic data, whereas Phase I trials assess safety, tolerability, and dosage of the drug.
- Phase 0 trials are exploratory and do not require traditional safety and efficacy endpoints, unlike Phase I trials.
- Phase 0 trials are typically shorter in duration and less costly compared to Phase I trials.
Phase I Clinical Trials
Phase I clinical trials are typically the first step in testing a new drug or treatment in humans after successful animal studies. These trials focus on assessing the safety and dosage levels of the treatment, rather than its effectiveness.
Characteristics of Phase I Clinical Trials
- Phase I trials involve a small group of healthy volunteers or patients with the specific condition being targeted by the treatment.
- The primary goal is to determine the safety and tolerability of the treatment at different dose levels.
- These trials are usually conducted in specialized research centers under close medical supervision.
Safety Assessments in Phase I Trials
- Regular monitoring of vital signs, blood tests, and physical examinations are conducted to assess any potential side effects.
- Adverse events are closely monitored and reported to ensure the safety of participants.
- Dose escalation may occur based on the safety data collected during the trial.
Sample Size and Duration of Phase I Trials
- Phase I trials typically involve a small number of participants, usually ranging from 20 to 80 individuals.
- The duration of these trials can vary but commonly lasts from a few weeks to several months, depending on the study design and objectives.
Dosages and Administration Methods in Phase I Trials
- Participants in Phase I trials are usually divided into groups that receive different doses of the treatment to determine the optimal dosage with the least side effects.
- The treatment is administered in a controlled setting, either orally, intravenously, or through other appropriate routes depending on the drug formulation.
Phase II Clinical Trials
Phase II clinical trials play a crucial role in the drug development process, building on the initial safety data obtained in Phase I trials. These trials aim to assess the effectiveness of a drug or treatment and further evaluate its safety profile.
Primary Objectives of Phase II Clinical Trials
Phase II trials primarily focus on determining the optimal dose of the drug, evaluating its efficacy in treating the targeted condition, and gathering more information on its safety profile before proceeding to larger Phase III trials.
Patient Population in Phase II Trials
The patient population involved in Phase II trials typically includes individuals diagnosed with the specific disease or condition being targeted by the investigational drug. These patients often have characteristics that make them suitable for testing the drug’s effectiveness and safety.
Design and Endpoints of Phase II Trials
Phase II trials are designed to assess the drug’s effectiveness through various endpoints such as tumor response rates, disease progression, or symptom improvement. These trials may also explore different dosages or treatment regimens to identify the most promising approach for further evaluation.
Examples of Diseases Studied in Phase II Trials
Some common diseases studied in Phase II clinical trials include:
- Various types of cancer, such as lung cancer, breast cancer, or leukemia
- Autoimmune diseases like rheumatoid arthritis or multiple sclerosis
- Infectious diseases such as HIV/AIDS or hepatitis
- Neurological disorders like Alzheimer’s disease or Parkinson’s disease
Phase III Clinical Trials
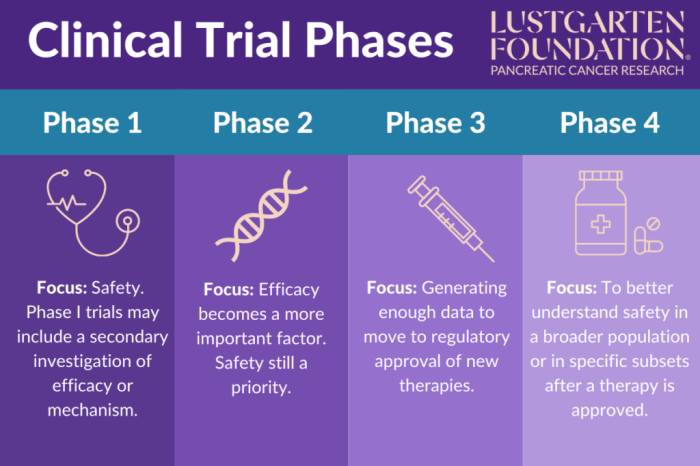
Phase III clinical trials are a crucial stage in the drug development process, where the focus shifts to evaluating the effectiveness of the new treatment in comparison to existing standard treatments. These trials are conducted on a larger scale and involve a more diverse group of participants to gather more comprehensive data on the drug’s safety and efficacy.
Scale and Scope of Phase III Clinical Trials
Phase III clinical trials typically involve hundreds to thousands of participants across multiple sites to ensure the generalizability of the results. These trials aim to confirm the findings from earlier phases, determine the optimal dosage, and assess the overall benefit-risk ratio of the new treatment.
Randomized and Controlled Aspects of Phase III Trials
Phase III trials are randomized, meaning participants are assigned to either the experimental group receiving the new treatment or the control group receiving standard treatment or a placebo. This random assignment helps eliminate bias and ensures reliable results. Additionally, Phase III trials are controlled, with researchers closely monitoring and comparing the outcomes between the two groups.
Regulatory Requirements and Considerations in Phase III Trials
Regulatory bodies like the FDA and EMA have strict requirements for Phase III clinical trials to ensure patient safety and data integrity. Researchers must adhere to Good Clinical Practice (GCP) guidelines, submit detailed study protocols, and report any adverse events promptly. Regulatory approval is necessary before initiating Phase III trials.
Significance of Phase III Trials in Drug Development
Phase III trials play a crucial role in determining the overall effectiveness and safety profile of a new treatment. Positive results from Phase III trials are often required for regulatory approval and eventual market authorization. These trials provide vital data for healthcare providers, regulators, and patients when making informed decisions about the use of the new treatment.
Concluding our exploration of Clinical Trials Phases Explained, we have navigated through the complexities of each phase, highlighting their unique contributions to advancing healthcare and pharmaceuticals.
Essential Questionnaire
What are the different phases of clinical trials?
The phases include Phase 0, Phase I, Phase II, and Phase III, each with specific objectives and criteria.
How are volunteers important in clinical trials?
Volunteers are crucial in advancing medical research by participating in clinical trials, helping to evaluate the safety and efficacy of new treatments.
What is the significance of Phase III clinical trials?
Phase III trials play a vital role in drug development, assessing the effectiveness and safety of new treatments on a larger scale before regulatory approval.





