Embark on your journey to learn ‘How to Participate in Clinical Trials’ as we delve into the world of medical research, uncovering the steps and requirements for those eager to contribute to scientific advancements.
Discover the phases, preparation, and post-trial aspects that are crucial for anyone considering involvement in clinical trials.
Understanding Clinical Trials
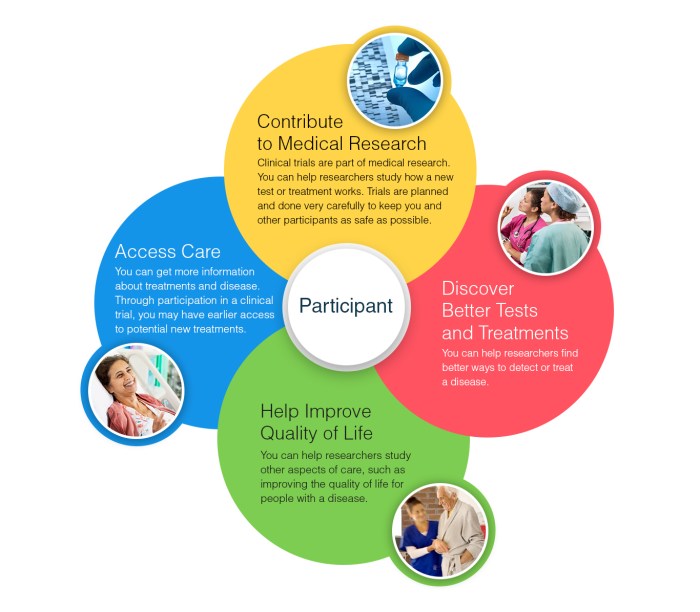
Clinical trials are research studies that test how well new medical treatments work in humans. These trials are crucial in advancing medical knowledge and improving patient care. By participating in clinical trials, individuals can contribute to the development of new treatments and potentially benefit from innovative therapies.
Phases of Clinical Trials
Clinical trials are typically divided into four phases:
Phase 1
These trials involve a small group of healthy volunteers to assess the safety and dosage of a new treatment.
Phase 2
In this phase, the focus shifts to testing the effectiveness of the treatment in a larger group of patients with the targeted condition.
Phase 3
These trials involve a larger number of participants to further evaluate the treatment’s effectiveness, monitor side effects, and compare it to existing treatments.
Phase 4
After the treatment is approved by regulatory authorities, phase 4 trials continue to monitor its long-term safety and effectiveness in a real-world setting.
Examples of Successful Treatments
Clinical trials have led to the development of numerous successful treatments, including:
- The HPV vaccine, which has significantly reduced the incidence of cervical cancer.
- Statins, a widely used medication for lowering cholesterol and reducing the risk of heart disease.
- Immunotherapy drugs, which have revolutionized cancer treatment by harnessing the body’s immune system to fight cancer cells.
Finding Clinical Trials to Participate In

Finding ongoing clinical trials to participate in can be a crucial step in contributing to medical research and potentially accessing innovative treatments. Here are some tips on how to search for clinical trials, the criteria for participating, and the role of informed consent in the process.
Tips for Finding Clinical Trials
- Utilize online databases: Websites like ClinicalTrials.gov and CenterWatch.com provide comprehensive listings of clinical trials across various medical conditions.
- Consult healthcare providers: Physicians or healthcare professionals may be aware of clinical trials that are suitable for your condition and can provide guidance.
- Join patient advocacy groups: Organizations dedicated to specific health conditions often share information about ongoing clinical trials and support participation.
Criteria for Participating in Clinical Trials
- Age, gender, and specific medical condition: Clinical trials often have specific criteria regarding the age, gender, and health status of participants.
- Health status and medical history: Participants may need to meet certain health requirements or have particular medical histories to be eligible for a trial.
- Willingness to comply with study protocols: Participants must be willing to follow the guidelines and procedures Artikeld in the clinical trial.
Role of Informed Consent
Informed consent is a critical aspect of clinical trial participation, where participants are provided detailed information about the study, including its purpose, procedures, potential risks, and benefits. Participants must fully understand the information provided before voluntarily agreeing to participate in the trial. This process ensures that individuals are making an informed decision about their involvement in the research.
Preparing for Participation
Before participating in a clinical trial, there are important steps to take to ensure you are fully prepared. It is crucial to consult with your healthcare provider, understand the potential risks and benefits, and make an informed decision.
Consulting with Healthcare Providers
Prior to joining a clinical trial, it is essential to consult with your healthcare provider. They can provide valuable insight into whether participating in a trial is the right decision for you based on your medical history, current health status, and treatment options. Your healthcare provider can also help you understand the purpose of the trial, the procedures involved, and any potential impact on your health.
Considering Risks and Benefits
When preparing to participate in a clinical trial, it is important to carefully consider the potential risks and benefits. Clinical trials may offer access to cutting-edge treatments and healthcare advancements, but they also come with potential risks such as side effects, adverse reactions, or unforeseen complications. It is crucial to weigh these risks against the potential benefits and make an informed decision based on your individual circumstances.
During the Clinical Trial
During a clinical trial, participants can expect to be closely monitored by healthcare professionals to ensure their safety and the integrity of the trial results. It is crucial for participants to follow the trial protocol diligently and attend all scheduled visits as per the instructions provided.
Monitoring Process and Role of Healthcare Professionals
- Participants will be regularly monitored by healthcare professionals to assess their health status and any potential side effects of the treatment being tested.
- Healthcare professionals will closely observe participants’ adherence to the trial protocol and provide guidance on any necessary adjustments.
- Monitoring may involve physical examinations, laboratory tests, imaging studies, and other measurements to track the progress of the participant throughout the trial.
Importance of Following Trial Protocol and Attending Scheduled Visits
- Adhering to the trial protocol is crucial for maintaining the validity and reliability of the trial results, ensuring that the data collected is accurate and meaningful.
- Attending scheduled visits allows healthcare professionals to closely monitor participants’ health, provide necessary support, and address any concerns or questions that may arise during the trial.
- Missing scheduled visits or deviating from the trial protocol without prior approval can impact the trial’s outcomes and may lead to the participant’s disqualification from the study.
After the Clinical Trial

Upon completion of a clinical trial, there are several important aspects to consider. This stage marks the end of the trial itself but not necessarily the end of your involvement in the study.
Follow-up Care and Long-term Monitoring
After the clinical trial ends, participants may receive follow-up care to monitor their health and well-being. This could involve regular check-ups, tests, or consultations with healthcare providers to ensure that any effects of the trial intervention are properly managed. Long-term monitoring may be necessary to track any potential long-lasting impacts or benefits from the trial.
- Follow-up Care: Participants might continue to receive medical attention to address any ongoing health concerns or to monitor the lasting effects of the trial intervention.
- Long-term Monitoring: Regular assessments may be scheduled to evaluate the sustained effects of the treatment or intervention tested in the trial.
Sharing Feedback and Experiences
It is crucial for participants to share their feedback and experiences after participating in a clinical trial. This information can provide valuable insights for researchers, helping them improve future studies and treatments. By sharing your experiences, you contribute to the advancement of medical knowledge and the development of better healthcare options for others.
Sharing your feedback can lead to enhancements in the design and implementation of clinical trials, ultimately benefiting both current and future participants.
In conclusion, navigating the realm of clinical trials requires careful consideration and preparation. By understanding the process from start to finish, individuals can make informed decisions about their participation, ultimately contributing to the advancement of medical knowledge and treatments.
User Queries
What are the different phases of clinical trials?
The phases include testing on small groups of people to determine safety (Phase 1), studying effectiveness and side effects (Phase 2), and comparing with existing treatments (Phase 3).
How can I search for ongoing clinical trials?
You can visit clinical trial databases, research hospitals, or consult with healthcare providers to find trials relevant to your condition.
What happens after a clinical trial ends?
After the trial, participants may receive follow-up care, be monitored for long-term effects, and are encouraged to share their feedback and experiences.




