
Delving into Clinical Trials for Cancer Treatment, this introduction immerses readers in a unique and compelling narrative. Clinical trials play a crucial role in revolutionizing cancer treatment, offering hope and cutting-edge solutions to patients worldwide.
Exploring the different phases, types, and ethical considerations surrounding clinical trials provides a comprehensive understanding of their impact on cancer research and patient care.
Overview of Clinical Trials
Clinical trials play a crucial role in the development of new cancer treatments, helping to determine the safety and effectiveness of these therapies. These trials involve rigorous testing in human subjects to evaluate the potential benefits and risks associated with the treatment.
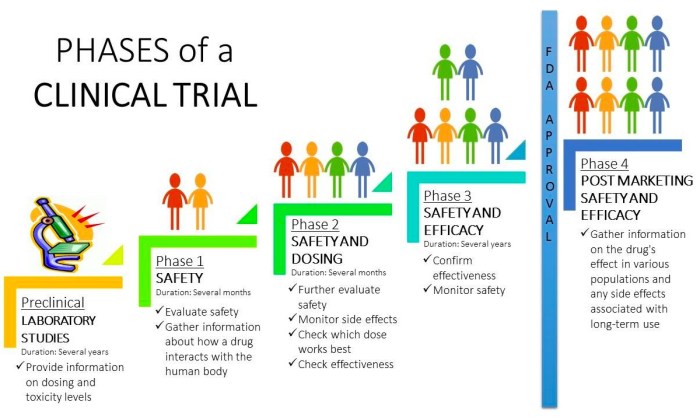
Phases of Clinical Trials
- Phase 1: In this initial phase, the focus is on determining the safety of the treatment and the appropriate dosage. A small group of participants is involved in these trials.
- Phase 2: This phase aims to further evaluate the safety and effectiveness of the treatment in a larger group of patients with the specific type of cancer being targeted.
- Phase 3: The treatment is compared to the standard treatment or a placebo in a large group of participants to confirm its effectiveness, monitor side effects, and collect additional information.
Importance of Clinical Trials in Advancing Cancer Treatment
Clinical trials are essential for advancing cancer treatment by providing valuable data on the efficacy and safety of new therapies. They help researchers and healthcare providers make informed decisions about the best treatment options for patients. Additionally, participating in clinical trials gives patients access to cutting-edge treatments that may not be available through standard care.
Types of Clinical Trials
Clinical trials for cancer treatment can be categorized into different types based on their specific objectives and methodologies. These types include treatment trials, prevention trials, diagnostic trials, and screening trials. Each type plays a crucial role in advancing cancer research and improving patient outcomes.
Treatment Trials
Treatment trials focus on evaluating new treatments, therapies, or medications for cancer patients. These trials aim to determine the effectiveness and safety of innovative approaches in treating cancer. For example, ongoing clinical trials may investigate the use of immunotherapy, targeted therapy, or combination treatments for specific types of cancer. By participating in treatment trials, patients can access cutting-edge treatments that may offer better outcomes than standard therapies.
Prevention Trials
Prevention trials are designed to explore interventions or strategies that can reduce the risk of developing cancer. These trials may involve the use of medications, lifestyle modifications, or vaccines to prevent cancer occurrence in high-risk individuals. Ongoing clinical trials in cancer prevention may study the impact of dietary supplements, exercise regimens, or chemopreventive agents on cancer risk reduction. Participation in prevention trials can provide valuable insights into effective cancer prevention strategies.
Diagnostic Trials
Diagnostic trials focus on developing and improving methods for early cancer detection and diagnosis. These trials aim to identify biomarkers, imaging techniques, or screening tests that can accurately detect cancer at its earliest stages. Ongoing clinical trials in cancer diagnostics may investigate the efficacy of liquid biopsies, molecular imaging, or artificial intelligence-based tools for early cancer detection. By participating in diagnostic trials, patients can contribute to the development of non-invasive and more precise diagnostic approaches.
Screening Trials
Screening trials aim to evaluate the effectiveness of screening programs in detecting cancer in asymptomatic individuals. These trials assess the impact of routine screening tests, such as mammograms, colonoscopies, or Pap smears, on early cancer detection and improved survival rates. Ongoing clinical trials in cancer screening may focus on optimizing screening protocols, evaluating new screening technologies, or assessing the cost-effectiveness of screening programs.
Participation in screening trials can help identify the most efficient and beneficial screening strategies for different types of cancer.
Informed Consent in Clinical Trials

In the context of clinical trials, informed consent refers to the process where participants are provided with all relevant information about the trial before agreeing to take part. This includes details about the purpose of the study, potential risks and benefits, procedures involved, and the participant’s rights.
Obtaining Informed Consent
The process of obtaining informed consent from participants in cancer treatment trials is crucial and follows strict guidelines. Researchers must ensure that participants fully understand the information provided before voluntarily agreeing to participate. This typically involves a detailed discussion with the participant, providing written materials for review, and allowing sufficient time for questions and clarification.
- Participants must be informed about the purpose of the trial, potential risks and benefits, and any alternative treatment options available.
- Researchers must explain the procedures involved, including any tests, medications, or interventions that will be administered during the trial.
- Participants have the right to withdraw from the trial at any time without penalty or impact on their medical care.
- Informed consent must be documented in writing, and participants are encouraged to seek advice from their healthcare provider or a trusted individual before making a decision.
Ethical Considerations
Informed consent in cancer treatment trials raises important ethical considerations. It is essential to ensure that participants are fully aware of the potential risks and benefits of the trial, as well as any uncertainties or unknown factors. Researchers must prioritize the well-being and autonomy of participants, respecting their right to make an informed decision about their participation.
It is crucial to uphold the principles of autonomy, beneficence, and justice when obtaining informed consent in clinical trials.
Patient Recruitment and Selection
Patient recruitment for cancer treatment clinical trials involves various strategies to identify and enroll suitable participants. These individuals play a crucial role in advancing medical research and improving cancer treatment options for the future.
Recruitment Strategies
- Medical Referrals: Healthcare providers may recommend eligible patients for clinical trials based on their medical history and treatment needs.
- Community Outreach: Collaborating with local organizations and support groups to raise awareness about clinical trials and encourage participation.
- Online Platforms: Utilizing websites and social media to reach a wider audience and connect with potential participants who may be interested in joining a clinical trial.
Selection Criteria
- Patient Health Status: Participants must meet specific health criteria to ensure their safety and the validity of the trial results.
- Age and Gender: Some clinical trials may have specific age or gender requirements based on the nature of the treatment being studied.
- Medical History: Patients with certain medical conditions or treatments may be excluded from participating in a clinical trial to prevent potential complications.
Challenges and Strategies
- Low Awareness: Many patients may not be aware of clinical trial opportunities or the benefits of participating. Educating the community and providing clear information can help address this challenge.
- Inclusion Criteria: Stringent eligibility criteria in some trials may limit the pool of potential participants. Adjusting criteria or expanding recruitment efforts can help overcome this obstacle.
- Logistical Barriers: Factors like distance to the research site, transportation issues, and scheduling conflicts can hinder patient recruitment. Offering support services and flexibility can improve recruitment rates.
Role of Placebos in Clinical Trials
The use of placebos in clinical trials plays a crucial role in evaluating the effectiveness of new cancer treatments and ensuring the reliability of the results obtained.
Use of Placebos in Cancer Treatment Clinical Trials
In cancer treatment clinical trials, placebos are often used as a control group to compare the effects of the new treatment against no treatment or standard treatment. This helps researchers determine the true impact of the experimental treatment on patients’ outcomes by eliminating biases and placebo effects.
Ethical Considerations of Using Placebos in Clinical Research
The use of placebos in clinical research raises ethical concerns, especially in the context of cancer treatment where patients are in urgent need of effective therapies. Researchers must ensure that the potential benefits of using placebos outweigh the risks to the participants, and informed consent is obtained from all participants before the trial begins.
How Placebos Help in Evaluating the Effectiveness of New Cancer Treatments
Placebos play a vital role in clinical trials by providing a baseline for comparison. They help researchers differentiate between the actual effects of the new treatment and any psychological or subjective responses from patients. By including a placebo group in the study design, researchers can accurately assess the true impact of the treatment and determine its efficacy in treating cancer.
In conclusion, Clinical Trials for Cancer Treatment are a cornerstone of progress in oncology, driving innovation and improving outcomes for those battling the disease. By participating in these trials, patients contribute to a future with more effective and personalized cancer treatments.
Question & Answer Hub
How are patients selected for clinical trials?
Patients are selected based on specific criteria set by the trial protocol, which may include factors such as the type and stage of cancer, overall health, and previous treatments.
What is the role of placebos in cancer treatment trials?
Placebos are used to evaluate the effectiveness of new treatments by comparing the results of the experimental treatment group with those receiving a placebo. They help researchers determine the true impact of the treatment.
Why is informed consent important in clinical trials?
Informed consent ensures that participants understand the risks and benefits of the trial, as well as their rights. It is essential for ethical conduct and respects the autonomy of individuals involved in the research.




