
Delving into Clinical Trials and Informed Consent, this introduction immerses readers in a unique and compelling narrative, with casual formal language style that is both engaging and thought-provoking from the very first sentence.
As we embark on this exploration, we will uncover the fundamental aspects of clinical trials and the critical significance of informed consent in medical research.
CLINICAL TRIALS
Clinical trials are research studies conducted to evaluate the safety and effectiveness of new medical treatments, interventions, or procedures. These trials play a crucial role in advancing medical knowledge and improving patient care by testing the efficacy of potential treatments before they are widely available.
Phases of Clinical Trials
- Phase 1: In this phase, the focus is on evaluating the safety of a new treatment and determining the appropriate dosage. A small group of healthy volunteers is usually involved.
- Phase 2: This phase involves a larger group of patients to assess the effectiveness of the treatment and further evaluate its safety.
- Phase 3: The treatment is tested on a larger group of patients to confirm its effectiveness, monitor side effects, and compare it to existing treatments.
- Phase 4: Post-marketing studies are conducted after the treatment has been approved to gather additional information on its risks, benefits, and optimal use.
Role of Participants, Researchers, and Sponsors
- Participants: Individuals who volunteer to take part in clinical trials play a crucial role in advancing medical science. They follow the trial protocol, attend study visits, and provide valuable data to researchers.
- Researchers: Medical professionals oversee clinical trials, design the study protocols, collect and analyze data, and ensure the safety and well-being of participants.
- Sponsors: Organizations or companies that fund and oversee clinical trials are known as sponsors. They play a key role in ensuring the trial is conducted ethically and in compliance with regulations.
Importance of Clinical Trials
Clinical trials are essential for developing new treatments, medications, and medical devices. They help determine the safety and efficacy of interventions, identify potential side effects, and provide evidence to support regulatory approval. By participating in clinical trials, individuals contribute to the advancement of healthcare and have the opportunity to access cutting-edge treatments before they are widely available.
INFORMED CONSENT
Informed consent is a fundamental ethical principle in clinical trials that ensures participants are fully aware of the risks and benefits before agreeing to take part. It is crucial for protecting the rights and welfare of individuals involved in research studies.
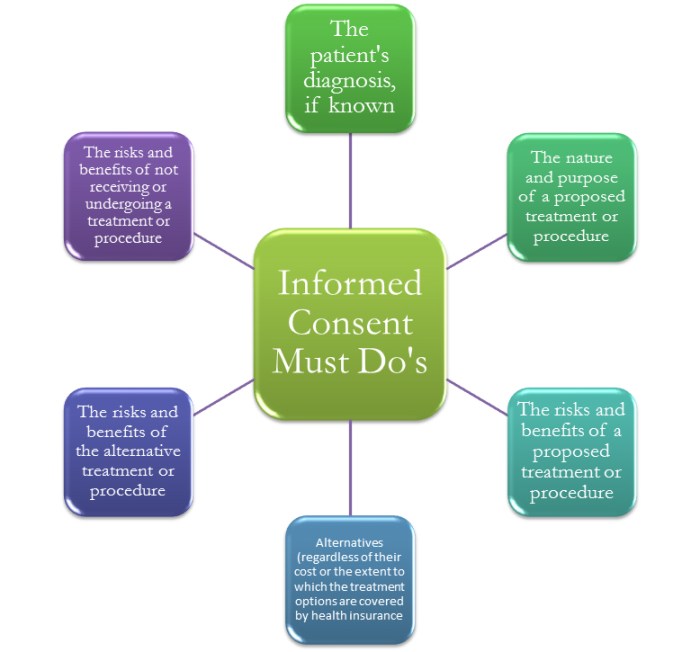
Key Elements of Informed Consent
- The purpose of the study and its procedures
- Potential risks and benefits of participation
- Alternative treatment options
- The voluntary nature of participation
- Contact information for questions and concerns
Protection of Participants’ Rights
By obtaining informed consent, participants have the autonomy to make an informed decision about their involvement in a clinical trial. This process ensures that individuals are not coerced or misled into participating and have the necessary information to make a voluntary choice.
Challenges and Ethical Considerations
- Language barriers and comprehension issues
- Vulnerable populations requiring extra safeguards
- The influence of financial incentives on decision-making
- Ensuring ongoing consent throughout the trial
- Balancing the need for informed consent with the urgency of certain research studies
INFORMED CONSENT PROCESS

In the informed consent process of clinical trials, several crucial steps are involved to ensure that participants fully understand the study’s procedures, risks, and benefits before agreeing to take part.
Steps in Obtaining Informed Consent
- Introduction: The healthcare professional explains the purpose of the study, procedures involved, and the participant’s role.
- Information Disclosure: Detailed information about the study, including risks, benefits, and alternatives, is provided to the participant.
- Opportunity for Questions: Participants are given the chance to ask questions and seek clarification on any aspects of the study.
- Voluntary Agreement: The participant voluntarily agrees to take part in the study without any coercion or pressure.
- Documentation: The informed consent form is signed by both the participant and the healthcare professional, acknowledging understanding and agreement.
Information Provided to Participants
- Study Purpose: Explanation of the research objectives and what the study aims to achieve.
- Procedures: Detailed description of the study procedures, including any tests, treatments, or interventions involved.
- Risks and Benefits: Clear Artikel of potential risks, benefits, and any available alternatives to participating in the study.
- Confidentiality: Assurance of data confidentiality and how participant information will be protected.
Role of Healthcare Professionals
- Clarifying Information: Healthcare professionals play a key role in ensuring participants understand complex medical information in simple terms.
- Answering Questions: Addressing participant questions and concerns to facilitate informed decision-making.
- Ensuring Voluntary Participation: Healthcare professionals must ensure that participants join the study willingly without any external pressure.
Best Practices for Obtaining Informed Consent
- Use Layperson’s Terms: Explain medical jargon in simple language to enhance participant comprehension.
- Encourage Questions: Create an open environment for participants to ask questions and seek clarification.
- Provide Ample Time: Allow participants sufficient time to review the information and make an informed decision.
- Document Understanding: Ensure that participants understand the study details before signing the consent form.
LEGAL AND ETHICAL ASPECTS
When it comes to clinical trials and informed consent, there are crucial legal and ethical aspects that must be considered to protect the rights and safety of participants.
Legal Requirements and Regulations
Legal requirements and regulations surrounding informed consent in clinical trials vary from country to country. However, there are some common principles that typically govern this process. In general, informed consent must be voluntary, informed, and given by individuals with the capacity to understand the information provided. Researchers must ensure that participants are fully aware of the risks, benefits, and alternatives to participation before obtaining their consent.
Ethical Considerations with Vulnerable Populations
Obtaining informed consent from vulnerable populations, such as children, elderly individuals, or those with cognitive impairments, requires additional ethical considerations. Researchers must take extra precautions to ensure that these individuals are not coerced or manipulated into participating in clinical trials. Special measures need to be implemented to safeguard their rights and well-being during the informed consent process.
Comparison of Informed Consent Process Across Regions
The informed consent process can vary significantly across different countries and regions due to cultural norms, legal frameworks, and healthcare practices. Some regions may have stricter regulations in place to protect participants, while others may have more lenient requirements. Understanding these differences is crucial for conducting ethical and compliant clinical trials on a global scale.
Implications of Failing to Obtain Proper Informed Consent
Failing to obtain proper informed consent in clinical trials can have serious consequences, both legally and ethically. It can lead to harm or exploitation of participants, damage to the reputation of the research institution, and invalidation of study results. Researchers must prioritize the informed consent process to uphold ethical standards and ensure the validity and integrity of their research.
In conclusion, the journey through Clinical Trials and Informed Consent sheds light on the intricate process of research ethics and participant protection, emphasizing the pivotal role they play in advancing healthcare innovation.
Questions and Answers
What are the different phases of clinical trials?
The phases include testing for safety, dosage, efficacy, and post-market surveillance.
How does informed consent protect participants?
It ensures participants are fully aware of the risks, benefits, and procedures involved in the trial.
What legal requirements govern informed consent?
Laws mandate that participants voluntarily consent after understanding all relevant information.




