Exploring the world of Clinical Trials for Alzheimer’s Disease, this introduction captivates readers with an intriguing narrative, providing essential information in a captivating manner.
The following paragraph will delve deeper into the various aspects of clinical trials for Alzheimer’s disease, shedding light on their importance and impact.
Introduction to Clinical Trials for Alzheimer’s Disease
Clinical trials play a crucial role in advancing research for Alzheimer’s disease, a progressive neurological disorder that affects millions worldwide. These trials are essential for testing new treatments, interventions, and prevention strategies to combat this devastating condition.
The Importance of Clinical Trials in Alzheimer’s Research
Alzheimer’s disease currently has no cure, making it vital to explore new avenues for potential treatments. Clinical trials provide researchers with the opportunity to evaluate the effectiveness and safety of experimental drugs, therapies, and approaches in a controlled setting.
Significance of Finding Effective Treatments
Finding effective treatments through clinical trials can significantly improve the quality of life for individuals living with Alzheimer’s disease. It can help slow down the progression of symptoms, delay cognitive decline, and enhance overall well-being.
General Process of Conducting Clinical Trials
- Identification of Research Participants: Individuals meeting specific criteria are recruited to participate in the trial.
- Randomization: Participants are randomly assigned to either the experimental group receiving the new treatment or the control group receiving a placebo or standard care.
- Monitoring and Data Collection: Researchers track participants’ progress, gather data on outcomes, and assess the safety and efficacy of the intervention.
- Analysis and Results: The collected data is analyzed to determine the treatment’s effectiveness, potential side effects, and overall impact on Alzheimer’s disease.
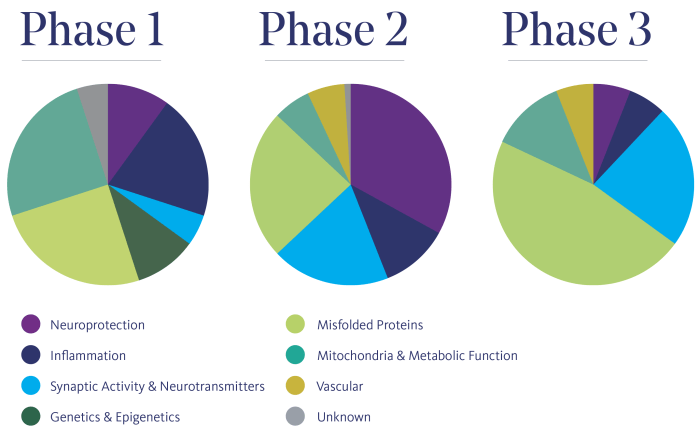
Types of Clinical Trials for Alzheimer’s Disease
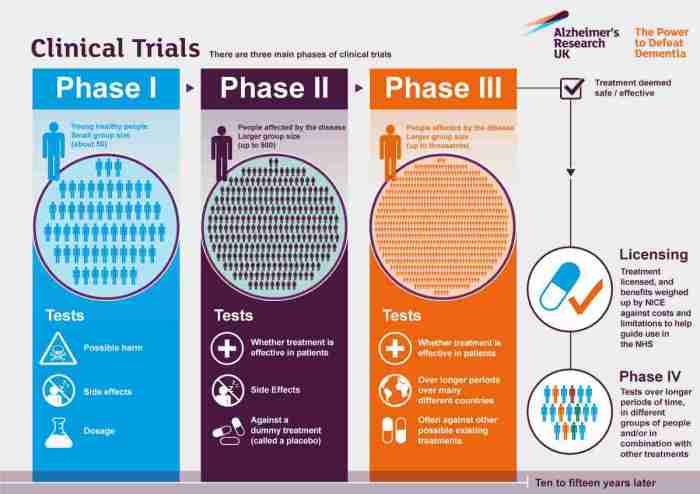
Clinical trials for Alzheimer’s disease are conducted in different phases, each with specific goals and methodologies. These phases are crucial in determining the safety and effectiveness of potential treatments for Alzheimer’s. Let’s explore the different phases of clinical trials in Alzheimer’s disease.
Phase I Clinical Trials
Phase I clinical trials are the first step in testing a new treatment or intervention in humans. The main goal of Phase I trials is to evaluate the safety and tolerability of the treatment in a small group of participants. Researchers also determine the appropriate dosage for further testing. These trials usually involve healthy volunteers or individuals with early-stage Alzheimer’s disease.
The methodologies used in Phase I trials focus on closely monitoring participants for any potential side effects and assessing how the treatment is metabolized in the body.
Phase II Clinical Trials
Phase II clinical trials involve a larger group of participants compared to Phase I trials. The primary goal of Phase II trials is to further evaluate the safety of the treatment and to assess its effectiveness in treating Alzheimer’s disease. Researchers also aim to gather preliminary data on the treatment’s efficacy and potential side effects. The methodologies in Phase II trials include randomized controlled trials to compare the treatment with a placebo or standard treatment.
These trials provide valuable insights into the treatment’s potential benefits and risks.
Phase III Clinical Trials
Phase III clinical trials are the final step before a potential treatment can be approved for use in patients. These trials involve a large number of participants across multiple sites. The main goals of Phase III trials are to confirm the treatment’s effectiveness, monitor side effects, and compare it to existing treatments. The methodologies used in Phase III trials include double-blind, randomized controlled trials to ensure unbiased results.
Phase III trials provide critical data on the treatment’s overall benefits and risks.
Phase IV Clinical Trials
Phase IV clinical trials, also known as post-marketing studies, occur after a treatment has been approved and is available to the public. The primary goal of Phase IV trials is to monitor the treatment’s long-term safety and effectiveness in a larger population. These trials help identify rare side effects or interactions that may not have been apparent during earlier phases.
The methodologies in Phase IV trials focus on real-world data collection and analysis to assess the treatment’s performance in a broader patient population.
Participant Recruitment in Clinical Trials
Recruiting participants for Alzheimer’s clinical trials is a crucial aspect of advancing research in the field. It involves reaching out to individuals who may be willing to participate in studies aimed at finding new treatments or interventions for Alzheimer’s disease.
Strategies for Recruiting Participants
- Utilizing medical centers and hospitals with specialized Alzheimer’s clinics to reach potential participants.
- Engaging with local communities through informational sessions, support groups, and outreach events.
- Collaborating with healthcare providers to identify eligible individuals who may be interested in participating.
- Utilizing online platforms and social media to raise awareness about clinical trials and recruit participants.
Criteria for Participant Enrollment
- Participants need to meet specific age requirements, usually adults over a certain age range.
- Individuals must have a diagnosis of Alzheimer’s disease or mild cognitive impairment.
- Participants should be willing and able to commit to the study protocols and follow-up visits.
- Some trials may have additional criteria related to medical history, current medications, or overall health status.
Challenges in Participant Recruitment
- Stigma associated with Alzheimer’s disease may deter individuals from participating in clinical trials.
- Difficulty in reaching diverse populations, including minorities and individuals in rural areas, can impact recruitment efforts.
- Concerns about the potential risks and side effects of experimental treatments may discourage participation.
- Logistical challenges such as transportation to study sites and scheduling conflicts can pose barriers to recruitment.
Ethical Considerations in Alzheimer’s Clinical Trials

When conducting clinical trials for Alzheimer’s disease, researchers must adhere to strict ethical principles to ensure the safety and well-being of participants.
Informed consent is a critical component of Alzheimer’s clinical trials, as it ensures that participants fully understand the purpose of the study, potential risks and benefits, and their rights as research subjects.
Researchers may encounter ethical dilemmas during these trials, such as balancing the need for scientific advancement with the protection of participants’ rights and safety.
Importance of Informed Consent
Informed consent is essential in Alzheimer’s clinical trials to protect the autonomy and rights of participants. It ensures that individuals have the necessary information to make an informed decision about participating in the study.
Ethical Dilemmas in Alzheimer’s Clinical Trials
Researchers may face ethical dilemmas when determining the appropriate level of risk for participants in Alzheimer’s clinical trials. Balancing the potential benefits of the study with the safety and well-being of participants can be challenging.
Another ethical dilemma researchers may encounter is ensuring that vulnerable populations, such as individuals with advanced Alzheimer’s disease, are not exploited or subjected to unnecessary risks during the trial.
In conclusion, this discussion has highlighted the critical role of clinical trials in advancing Alzheimer’s research and the ongoing quest for effective treatments.
General Inquiries
What are the common side effects participants may experience in Alzheimer’s clinical trials?
Common side effects may include headaches, nausea, fatigue, and dizziness, but each trial may vary in its specific side effect profile.
How long do clinical trials for Alzheimer’s disease typically last?
The duration of clinical trials can vary, but they usually last from several months to a few years, depending on the phase and goals of the study.




