Exploring the realm of Clinical Trials for Chronic Conditions, this introduction delves into the vital role they play in shaping medical advancements. From patient recruitment to data analysis, this overview highlights the critical aspects of conducting trials for chronic illnesses.
Introduction to Clinical Trials
Clinical trials are research studies conducted to evaluate the safety and effectiveness of new medical treatments, interventions, or medications on human volunteers. These trials play a crucial role in advancing medical knowledge and improving patient care by providing evidence-based data on the benefits and risks of potential treatments.
The Importance of Clinical Trials in Advancing Medical Research
Clinical trials are essential in the field of medicine as they help researchers and healthcare professionals to:
- Assess the safety and efficacy of new treatments
- Compare new treatments with existing standard treatments
- Identify potential side effects and risks of treatments
- Understand how treatments work in different populations
- Contribute to the development of evidence-based medical guidelines
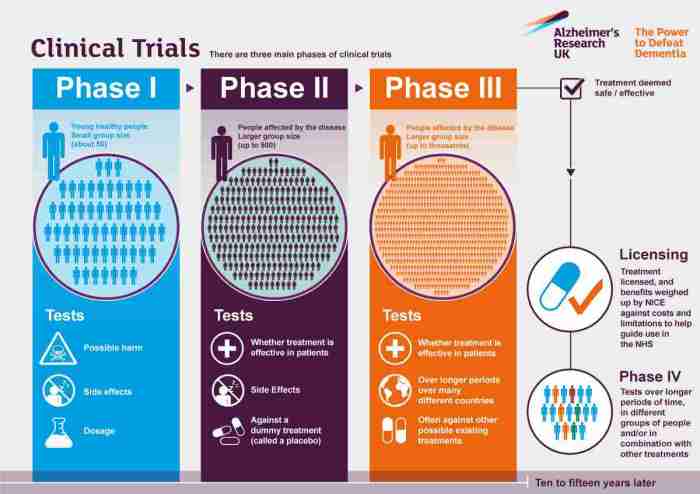
The Different Phases of Clinical Trials
Clinical trials are typically conducted in four phases, each serving a specific purpose in the research process:
- Phase 1: These trials involve a small group of healthy volunteers to evaluate the safety and dosage of a new treatment.
- Phase 2: In this phase, the focus is on determining the efficacy of the treatment in a larger group of patients with the condition of interest.
- Phase 3: These trials involve a larger group of patients to further evaluate the treatment’s effectiveness, monitor side effects, and compare it to standard treatments.
- Phase 4: After the treatment has been approved, phase 4 trials are conducted to monitor its long-term safety and effectiveness in real-world settings.
Types of Clinical Trials for Chronic Conditions
Clinical trials for chronic conditions can be categorized into different types based on their design and purpose. Two main types of clinical trials are observational trials and interventional trials. Observational trials involve observing and monitoring participants without any intervention, while interventional trials involve testing new treatments or interventions.
Randomized Controlled Trials
Randomized controlled trials (RCTs) are a crucial type of clinical trial in studying chronic conditions. In RCTs, participants are randomly assigned to different groups, with one group receiving the experimental treatment and the other receiving a standard treatment or a placebo. This design helps to minimize bias and ensures the reliability of the results obtained. RCTs are considered the gold standard for evaluating the efficacy and safety of new treatments for chronic conditions.
Patient Recruitment and Selection

Recruiting patients for clinical trials on chronic conditions is a crucial step in the research process. It involves reaching out to potential participants who meet the specific criteria set for the trial.Identifying the right participants for these trials is essential to ensure the validity and reliability of the study results. Researchers typically use specific criteria to select participants based on factors such as age, gender, medical history, and the severity of the chronic condition being studied.
Criteria for Participant Selection
- Age range: Researchers may specify an age range to ensure that participants are within a certain demographic group.
- Medical history: Participants with a documented history of the chronic condition being studied may be preferred.
- Health status: Participants must meet certain health criteria to ensure they can safely participate in the trial.
- Severity of the condition: The severity of the chronic condition may determine eligibility for the trial.
Importance of Diverse Participant Selection
It is crucial to have a diverse pool of participants in clinical trials for chronic conditions to ensure that the results are applicable to a broader population. A diverse participant selection helps researchers understand how different demographic groups respond to treatments and interventions, leading to more effective and inclusive healthcare practices.
Ethical Considerations in Clinical Trials
When conducting clinical trials for chronic conditions, ethical guidelines play a crucial role in ensuring the safety and well-being of participants as well as the integrity of the research process.
Informed Consent Process
The informed consent process is a fundamental ethical requirement in clinical trials. It involves providing potential participants with comprehensive information about the trial, including its purpose, procedures, risks, and benefits. Participants must fully understand this information before voluntarily agreeing to participate.
Institutional Review Boards (IRBs)
Institutional Review Boards (IRBs) are independent committees tasked with reviewing and approving research protocols to ensure that they adhere to ethical standards. IRBs evaluate the risks and benefits of the trial, the informed consent process, and the protection of participants’ rights. Their primary role is to safeguard the welfare and rights of trial participants.
Data Collection and Analysis
In clinical trials for chronic conditions, the data collection and analysis process plays a crucial role in determining the effectiveness and safety of the treatment being studied.
Data Collection Methods
There are several methods used for collecting data in clinical trials for chronic conditions:
- Medical examinations and tests: Patients undergo specific medical examinations and tests to assess their condition before, during, and after the trial.
- Patient-reported outcomes: Patients provide information about their symptoms, quality of life, and overall well-being through questionnaires and interviews.
- Laboratory tests: Blood tests, imaging studies, and other laboratory tests are conducted to measure specific biomarkers or indicators related to the chronic condition.
Statistical Analysis Techniques
Statistical analysis techniques are employed to interpret the data collected during the clinical trial. These techniques help researchers determine the significance of the results and whether the treatment has a meaningful impact on patients with chronic conditions.
- Descriptive statistics: Describing the data using measures such as mean, median, and standard deviation.
- Inferential statistics: Drawing conclusions and making predictions about the population based on sample data.
- Hypothesis testing: Testing whether the observed differences between treatment groups are statistically significant.
Data Integrity and Accuracy
Ensuring data integrity and accuracy is paramount in clinical research to maintain the credibility and validity of the trial results. Any errors or inconsistencies in data collection and analysis can lead to misleading conclusions and potentially harm patients with chronic conditions.
“Garbage in, garbage out”
The quality of the data collected directly impacts the quality of the results obtained.
Challenges and Limitations

Clinical trials for chronic conditions come with their fair share of challenges and limitations. These can impact the effectiveness and efficiency of the trials, as well as the overall quality of the data collected.
Recruitment Challenges
- Difficulty in finding eligible participants due to strict inclusion and exclusion criteria.
- Lack of awareness about clinical trials among the target population.
- High dropout rates during the trial duration, affecting the completion of the study.
Logistical Limitations
- Constraints in terms of resources, including funding, facilities, and qualified personnel.
- Complexity in managing multi-site trials, leading to coordination challenges.
- Adherence to strict regulatory requirements and guidelines, which can slow down the progress of the trial.
Data Collection and Analysis Issues
- Incomplete or inaccurate data collection due to participant non-compliance or errors in documentation.
- Challenges in analyzing long-term data and interpreting outcomes in the context of chronic conditions.
- Lack of standardized measures and endpoints across different trials, affecting comparability.
Overcoming Challenges
- Improving community engagement and education to enhance participant recruitment.
- Utilizing technology for remote monitoring and data collection to overcome logistical barriers.
- Enhancing collaboration among research teams and institutions to streamline multi-site trials.
In conclusion, Clinical Trials for Chronic Conditions stand as a cornerstone of medical progress, offering insights into treatment efficacy and patient outcomes. By addressing challenges and upholding ethical standards, these trials pave the way for improved healthcare practices and patient care.
FAQ Resource
How are participants selected for clinical trials?
Participants are selected based on specific criteria set by researchers, ensuring the trial’s integrity and relevance to the study.
What is the role of Institutional Review Boards in clinical trials?
Institutional Review Boards oversee the ethical conduct of trials, safeguarding participants’ rights and ensuring research compliance with regulatory standards.
Why is data integrity crucial in clinical research?
Data integrity is vital for ensuring the accuracy and reliability of study findings, influencing treatment decisions and medical advancements.




