
Embark on a journey to uncover the world of clinical trials, exploring their definition, purpose, significance, and phases in a captivating manner.
Delve deeper into the realm of medical research as we unravel the complexities and importance of clinical trials.
What are Clinical Trials?
Clinical trials are research studies conducted in the medical field to evaluate the effectiveness and safety of new treatments, interventions, or medical devices on human subjects. These trials are essential in determining whether a new approach is better than existing ones or if it is safe for use.
Purpose of Conducting Clinical Trials
Clinical trials aim to gather data and evidence to determine the safety and efficacy of new treatments or interventions. They help researchers and healthcare professionals identify better ways to prevent, diagnose, and treat various medical conditions. By conducting these trials, medical advancements are made possible, leading to improved patient outcomes and quality of life.
Significance of Clinical Trials in Advancing Healthcare
- Clinical trials play a crucial role in developing new treatments and medications that can potentially save lives and improve health outcomes.
- They contribute to the expansion of medical knowledge and understanding of diseases, leading to better patient care and management.
- Through clinical trials, healthcare providers can offer patients access to cutting-edge treatments that may not be available through standard care.
- These trials also help in establishing evidence-based guidelines for clinical practice, ensuring that healthcare decisions are grounded in scientific research.
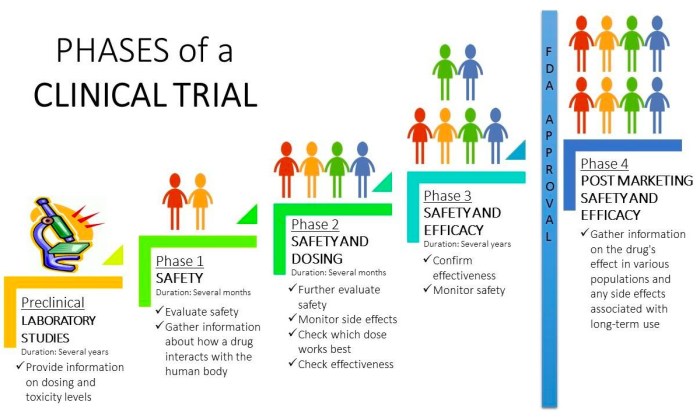
Different Phases of Clinical Trials
- Phase 1: Involves testing a new treatment on a small group of people to evaluate safety, dosage, and potential side effects.
- Phase 2: Expands the study to a larger group to further assess the treatment’s effectiveness and side effects.
- Phase 3: Compares the new treatment to existing standard treatments on a larger scale to determine its overall benefits and risks.
- Phase 4: Occurs after the treatment is approved for use and involves monitoring its long-term effects in the general population.
Types of Clinical Trials
Clinical trials are essential in advancing medical research and improving patient care. There are several types of clinical trials conducted to evaluate the safety and efficacy of new treatments. Let’s explore the different types and their characteristics.
Observational vs. Interventional Clinical Trials
Observational clinical trials involve observing participants in their usual environment without any intervention. Researchers collect data on health outcomes and analyze the relationships between various factors. In contrast, interventional clinical trials involve administering a specific treatment to participants to evaluate its effects.
Randomized vs. Non-Randomized Clinical Trials
Randomized clinical trials involve randomly assigning participants to different treatment groups, ensuring that each group has an equal chance of receiving the treatment. This helps eliminate bias and ensures the reliability of the results. Non-randomized clinical trials do not involve random assignment of participants to treatment groups.
Clinical Trial Designs
- Parallel Design: Participants are divided into different groups, and each group receives a different treatment. This design allows for comparison of the treatment outcomes between groups.
- Crossover Design: Participants receive multiple treatments in a sequential order, allowing for comparison within the same group over time.
- Factorial Design: Participants receive multiple treatments simultaneously, enabling researchers to study the effects of each treatment alone and in combination.
Participants in Clinical Trials

Clinical trials rely on the participation of individuals who volunteer to be part of research studies that test new treatments, interventions, or medical devices. These participants play a crucial role in advancing medical knowledge and improving healthcare outcomes.
Role of Participants in Clinical Trials
Participants in clinical trials are essential for researchers to gather data on the safety and efficacy of new treatments or interventions. They follow study protocols, receive the experimental treatment or standard care, and provide valuable feedback on their experiences throughout the trial.
Criteria for Selecting Participants in Clinical Trials
- Age, gender, and medical history: Researchers may have specific criteria regarding the demographics and health status of participants to ensure the study results are applicable to the target population.
- Medical condition: Participants must have the medical condition under investigation to assess the effectiveness of the treatment being studied.
- Informed consent: Individuals must provide informed consent after receiving detailed information about the study objectives, procedures, risks, and benefits.
Importance of Diversity in Clinical Trial Participants
- Diversity in clinical trial participants is crucial to ensure that research findings are applicable to a broader population and not limited to specific demographics.
- Including diverse participants can help identify how treatments may affect different groups based on factors such as age, race, ethnicity, or genetic background.
Ethical Considerations in Recruiting Participants for Clinical Trials
- Respect for autonomy: Participants have the right to make informed decisions about their involvement in clinical trials without coercion.
- Beneficence and non-maleficence: Researchers must prioritize the well-being of participants and minimize potential risks while maximizing benefits.
- Data confidentiality: Protecting the privacy and confidentiality of participants’ personal and medical information is essential to maintain trust and ethical standards.
Conducting Clinical Trials
Clinical trials are crucial for advancing medical knowledge and improving patient care. The process of conducting these trials involves several key steps to ensure accuracy, safety, and ethical standards are met.
Designing a Clinical Trial
- Define the research question and objectives of the trial.
- Determine the study population and eligibility criteria for participants.
- Develop a detailed protocol outlining the study procedures, interventions, and outcomes.
- Randomize participants into treatment groups to minimize bias.
- Plan for data collection, analysis, and interpretation.
Obtaining Regulatory Approval
- Submit the trial protocol to regulatory authorities for review.
- Demonstrate that the trial meets ethical and scientific standards.
- Address any feedback or concerns raised by regulatory agencies.
- Receive approval before initiating the trial.
Role of Institutional Review Boards (IRBs)
- IRBs are responsible for protecting the rights and welfare of trial participants.
- Review trial protocols to ensure they meet ethical standards.
- Monitor the conduct of the trial and address any issues that arise.
- Have the authority to approve, modify, or reject trial protocols.
Challenges Faced in Clinical Trials
- Recruitment of participants can be challenging, affecting the timeline and success of the trial.
- Adherence to the protocol by participants and researchers is crucial for reliable results.
- Data collection and analysis require meticulous attention to detail to minimize errors.
- Adverse events or unexpected outcomes must be promptly reported and managed.
In conclusion, the realm of clinical trials is a crucial aspect of advancing healthcare, paving the way for innovative treatments and medical breakthroughs.
FAQ Overview
What are the different phases of clinical trials?
The phases include Phase 0, Phase 1, Phase 2, Phase 3, and Phase 4, each serving a specific purpose in the research process.
How are participants selected for clinical trials?
Participants are selected based on specific criteria Artikeld by the researchers, ensuring the safety and efficacy of the trial.
What is the role of Institutional Review Boards (IRBs) in clinical trials?
IRBs oversee the ethical aspects of clinical trials, safeguarding the rights and well-being of participants.




